CD : 60102
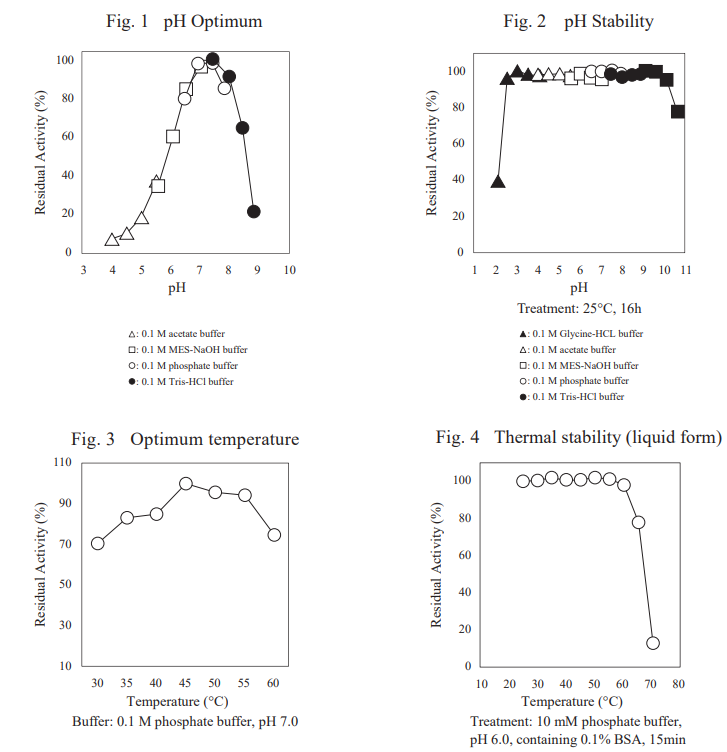
The enzyme is useful for the determination of D-glucose in clinical analysis. FADGDH-AD is an FAD-dependent glucose dehydrogenase with low reactivity toward maltose and xylose. It is one of the most stable glucose dehydrogenases for blood glucose measurement and is suitable for continuous glucose monitoring sensor.
| Origin | recombinant Aspergillus sojae |
|---|---|
| Systematic name | D-Glucose : acceptor 1-oxidoreductase |
| EC Number | 1.1.5.9 |
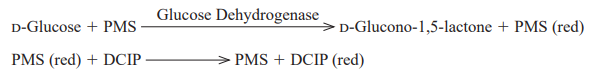
| Reaction formula | D-Glucose + acceptor →→→ D-Glucono-1,5-lactone + reduced acceptor |