Outline
| Product | Easy Plate (AC, EC, CC, SA, YM-R,EB) |
|---|---|
| Event date | 2025.05.08 |
Seminar Detail
| Venue | Madrid, Spain, Melia Avenida America |
|---|
Result contents
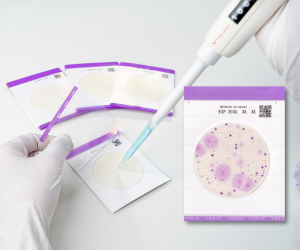
IAFP European Symposium 2025 – Technical Session 9
T9: Evaluation of Easy Plate YM-R™ for Enumerating Yeasts and Molds in Both High and Low Water Activity Foods
Introduction
Kikkoman Biochemifa Company will be presenting new validation data for Easy Plate YM-R™, a ready-to-use (RTU) culture medium developed for the rapid enumeration of yeasts and molds. The product enables results in 48 to 72 hours and is suitable for both high and low water activity foods. This presentation will take place as part of Technical Session 9 (T9) at the IAFP European Symposium 2025.
Purpose
The purpose of this study is to evaluate the performance of Easy Plate YM-R™ in alignment with AOAC Performance Tested Methods℠, MicroVal, and NordVal certification requirements, with particular focus on applied statistical methods for method validation.
Study Overview
Methodology:
Easy Plate YM-R™ was compared against ISO 21527-1/2 (2008) and BAM Chapter 18 (2001). Validation was conducted across diverse product categories including:
- Dairy
- Fruits and produce
- Multicomponent foods
- Bakery items
- Meat and poultry
- Dried foods
- Confectionery
- Pet foods
- Environmental swabs
Parameters assessed include:
- Relative trueness: 135 items using Bland-Altman plots
- Repeatability and accuracy: β-ETIs and confidence intervals (19 items)
- Selectivity: 50 inclusivity strains, 30 exclusivity strains Robustness, consistency, and stability: via ANOVA
Key Findings
- At 48 hours, Easy Plate YM-R™ demonstrated agreement with reference methods in three categories: produce, multicomponent foods, and environmental samples
- At 72 hours, consistent agreement was achieved across all tested food types, within ±0.5 log or recalculated acceptance limits
- Selectivity surpassed reference methods, with the medium supporting growth of all tested xerophilic and non-xerophilic inclusivity strains, and inhibiting all exclusivity strains
- Robustness testing indicated the importance of maintaining correct incubation temperature
- Consistency and long-term product stability were also confirmed
Conclusion
Easy Plate YM-R™ is a robust and time-saving alternative to traditional yeast and mold testing methods. It offers a simplified workflow, wide applicability across food matrices, and strong statistical validation. The method is especially valuable for manufacturers managing both high and low water activity products.
Presenters
- Shinichiro Sugiura – Primary Author and Presenter, Kikkoman Biochemifa Company
- Daiki Utsunomiya – Kikkoman Biochemifa Company
- Alice Foxall – Campden BRI
- Suzanne Jordan – Campden BRI
We welcome IAFP EU 2025 attendees to join Technical Session 9 (T9) to learn more about the validation and application of Easy Plate YM-R™ in microbiological testing. For additional information or to request a meeting during the event, please contact us through our [inquiry form] or speak with your local representative.
IAFP European Symposium 2025 – Technical Session 9
T9: Evaluation of Easy Plate YM-R™ for Enumerating Yeasts and Molds in Both High and Low Water Activity Foods
Introduction
Kikkoman Biochemifa Company will be presenting new validation data for Easy Plate YM-R™, a ready-to-use (RTU) culture medium developed for the rapid enumeration of yeasts and molds. The product enables results in 48 to 72 hours and is suitable for both high and low water activity foods. This presentation will take place as part of Technical Session 9 (T9) at the IAFP European Symposium 2025.
Purpose
The purpose of this study is to evaluate the performance of Easy Plate YM-R™ in alignment with AOAC Performance Tested Methods℠, MicroVal, and NordVal certification requirements, with particular focus on applied statistical methods for method validation.
Study Overview
Methodology:
Easy Plate YM-R™ was compared against ISO 21527-1/2 (2008) and BAM Chapter 18 (2001). Validation was conducted across diverse product categories including:
- Dairy
- Fruits and produce
- Multicomponent foods
- Bakery items
- Meat and poultry
- Dried foods
- Confectionery
- Pet foods
- Environmental swabs
Parameters assessed include:
- Relative trueness: 135 items using Bland-Altman plots
- Repeatability and accuracy: β-ETIs and confidence intervals (19 items)
- Selectivity: 50 inclusivity strains, 30 exclusivity strains Robustness, consistency, and stability: via ANOVA
Key Findings
- At 48 hours, Easy Plate YM-R™ demonstrated agreement with reference methods in three categories: produce, multicomponent foods, and environmental samples
- At 72 hours, consistent agreement was achieved across all tested food types, within ±0.5 log or recalculated acceptance limits
- Selectivity surpassed reference methods, with the medium supporting growth of all tested xerophilic and non-xerophilic inclusivity strains, and inhibiting all exclusivity strains
- Robustness testing indicated the importance of maintaining correct incubation temperature
- Consistency and long-term product stability were also confirmed
Conclusion
Easy Plate YM-R™ is a robust and time-saving alternative to traditional yeast and mold testing methods. It offers a simplified workflow, wide applicability across food matrices, and strong statistical validation. The method is especially valuable for manufacturers managing both high and low water activity products.
Presenters
- Shinichiro Sugiura – Primary Author and Presenter, Kikkoman Biochemifa Company
- Daiki Utsunomiya – Kikkoman Biochemifa Company
- Alice Foxall – Campden BRI
- Suzanne Jordan – Campden BRI
We welcome IAFP EU 2025 attendees to join Technical Session 9 (T9) to learn more about the validation and application of Easy Plate YM-R™ in microbiological testing. For additional information or to request a meeting during the event, please contact us through our [inquiry form] or speak with your local representative.
Related seminars
-

Webinar: From Conventional Ready-to-Use-Media to Easy Plate A Laboratory Perspective on Improving Microbiological Testing Efficiency
Overview:
Microbiological Laboratories are under increasing pressure to deliver accurate, reliable results while improving workflow efficiency. As testing demands evolve, many laboratories are reassessing traditional ready-to-use media solutions, including widely used film-based methods, and exploring tools that support greater speed, consistency, and automation. Join this technical weinar featuring SGS INLASA Guatemala, as they share their real world lboratory experience transitioning from conventional ready-to-use media to the Kikkoman Easy Plate Series, including implementation of the Colony Counting System (CCS).
What You’ll Learn:
In this session, attendees will gain practical insight into:
- The evaluation process when transitioning to Easy Plate
- Operational considerations for implementation in a microbiology laboratory
- Workflow improvements observed after adoption
- Measurable efficiency gains through the use of Easy Plate and CCS
- How modernization can support laboratory productivity while maintaining quality, compliance, and ISO 17025 standards
This webinar is designed to provide practical guidance for laboratories considering modernization of microbiological testing workflows.
Who Should Attend?
This webinar is ideal for:
- Microbiology laboratory managers QA/QC professionals
- Food safety professionals Laboratory technicians
- Testing laboratories and contract labs
- Food manufacturers considering workflow improvement
- Laboratories currently using ready-to-use media or film-based methods
-

- Event date
- 2025.09.03
- Product
- Easy Plate (AC, EC, CC, SA, YM-R,EB,AC-R)
- Venue
- Webinar
-

- Event date
- 2025.07.15
- Product
- Easy Plate (AC, EC, CC, SA, YM-R,EB,AC-R)
- Venue
- Webinar

